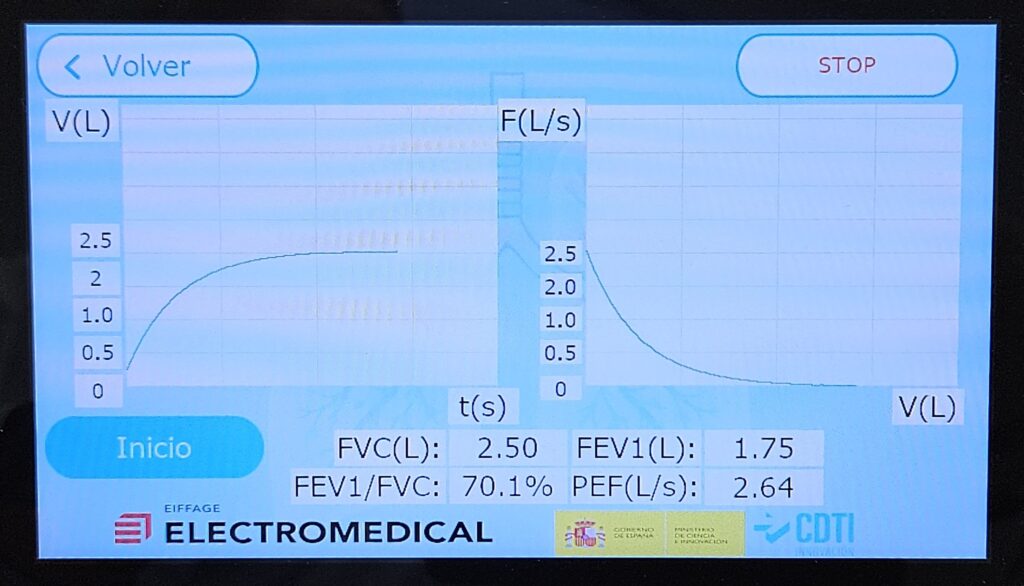
The R&D&I Department at Electromedical, a subsidiary of the Eiffage Energía Sistemas Group, has received a grant from the CDTI (Centre for Industrial Technological Development) to develop a technique for the dynamic calibration of spirometers used in the diagnosis of respiratory diseases.
According to the WHO, more than 3 billion people worldwide suffer from chronic respiratory diseases, which are the third leading cause of death among the elderly. The technique used to assess lung function is called spirometry and is performed using a medical device known as a spirometer.

SESPIRD™, a necessary and innovative project
Currently, volume (static) calibrations are performed using syringes with standardised volumes. However, in the case of flow (dynamic) calibrations, no affordable commercial solutions are available that enable this type of verification to be carried out on a widespread basis.
In response to this need, the SESPIRD™ pilot project has been developed: a reliable, user-friendly, and cost-effective dynamic calibration device, suitable for use in hospitals, clinics, and private healthcare providers.
The aim is to promote the broader adoption of this technique, thereby generating a positive societal impact by improving the diagnosis of lung diseases.
A technological challenge in dynamic airflow calibration
The project is based on two fundamental pillars. The first has focused on research to develop airflow delivery and control technology capable of meeting the requirements associated with dynamic calibration.
The second pillar has focused on the design, manufacture and validation of a device based on this technology, with the aim of ensuring controlled production costs that enable an affordable solution and facilitate its implementation.
The success of this project will make a decisive contribution to improving the measurement capability of spirometers, ensuring the accurate detection of exhaled airflow. Furthermore, it will enable access to
this technology for all types of users, from large hospitals to small clinics or mutual health insurance providers.
Design, development, and validation
Eiffage Electromedical, in collaboration with the IBV (Valencia Institute of Biomechanics), has advanced the SESPIRD™ project through three key phases: design, development, and validation.
During the design phase, the system’s requirements, functionalities, and components were defined, incorporating usability and safety criteria.
Subsequently, the technical development was carried out, including the manufacture and assembly of the prototype. Finally, the device was validated through testing to confirm its correct operation and reliability.
A technological need addressed
The prototype developed also meets three fundamental objectives: a cost-effective, high-precision design; the development of software for controlling and managing the calibrator; and the comprehensive validation of its operation.
This advancement addresses a significant technological need in the assessment of lung function and, consequently, in the detection and treatment of respiratory diseases such as asthma, chronic obstructive pulmonary disease (COPD), or cystic fibrosis, among others.
This progress reinforces the commitment of Eiffage Energía Sistemas, through its subsidiary Electromedical, to innovation in the field of electromedicine.